Professor James Economy's Group
|
|
[Polymer Blends] [Microelectronic Polymers]
[Liquid Crystalline
Polyesters] [Polyester Thermosetting Resins] Background
& Concepts When
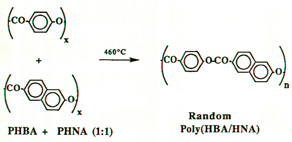
Professor Economy tried making liquid crystalline polymer
blends of polyhydroxybenzoic acid (pHBA) and
polynaphthoic acid (pHNA), back in 1987, he and his
researchers noticed that a random copolymer of HBA and
HNA was the result (Figure 1). This couldn't be readily
explained at first. Two homo-polymers of HBA and HNA
turning into a random copolymer of both was not expected.
As it turned out, interchain transesterification
reactions (ITR) were the cause of this randomization.
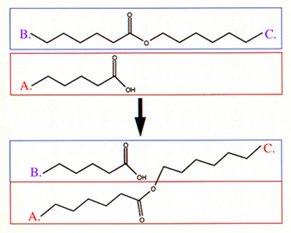
Figure 1:
Reaction Between Two Polymers Produced A Random Copolymer Interchain
Transesterification Reactions Interchain
transesterification reactions are intriguing. They are
reactions between two ester units across a polymer chain.
The ester-units swap back and forth between each chain.
It's this swapping mechanism which enabled the pure
homopolymers to end up as random chains. These reactions
are extremely fast, happening at 460 °C. Dr.
Economy saw an opportunity: "If these bonds are
being made between chains, could they be made between
chains that are on different plastic parts?" He and
his researchers bonded LCP coatings at various
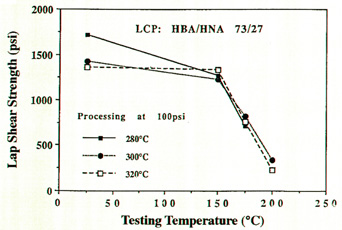
temperatures and tested them for lap-shear strengths.
Their results can be seen in Figure 2. Of importance is
the lap-shear strengths seen in the materials bonded at
280 °C. At this temperature, the LCP's used in the test
were not molten, hence, these parts were bonded to each
other in the solid-state.
Figure 2:
Lap-shear of LCP bonded substrates.
Figure 3:
Oligomer A had B. Solid-State
Consolidation ITs
Reactions can be exploited to bond polyesters while in
the solid-state. Both the Polyester Thermosets and the
Polyester LCP research we are currently conducting
utilize these unique reactions. Lap-shear testing has
been done on many resins. These samples fail cohesively,
not adhesively, which means the bond-strengths that can
be acheived through ITR bonding are as strong as the bulk
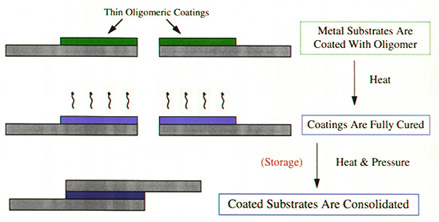
material. Lap-shear specimens utilizing our thermosetting
resins are made in the following way (Figure 4). First,
the oligomers are coated onto a substrate. The
thermosetting resins are then fully cured as separate
parts. These parts are then lapped and bonded together by
using heat and pressure.
Figure 4:
Solid state bonding using ITR.
Figure 5:
Schematic of novel approach to bonding through ITR. A
Recycleable Thermoset The
unique nature of the ITR bond-swapping gives our
polyesters one more interesting property not seen in
other thermosets. We can recycle these thermoset resins
and re-use them as oligomers either as an additive to
virgin resin or as a 100 % recycled resin. Our group in
the past has shown that these materials can be recycled
up to three times before any degredation in properties.
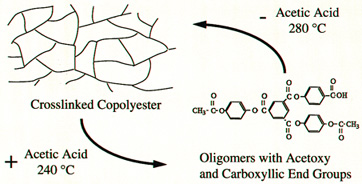
Figure 6 below shows the recycling process. The
condensate, acetic acid, is added back to the thermoset
and under heat and pressure, the system can be converted
back into a mixed-endgroup oligomer.
Figure 6:
Schematic of the recycling process. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||